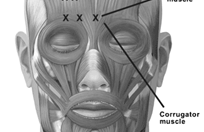
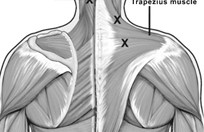
Botulin Toxin A for Chronic Migraine
Botulin Toxin A received FDA approval for the treatment of chronic migraine in October 2010. It is an injectable form of botulinum toxin used for preventing migraines in adults with chronic migraine (migraines that occur on 15 or more days per month for more than 3 months, which has the features of migraine headache on at least 8 days per month).
How Botulin Toxin A for migraine works
Botulin Toxin A works by blocking the release of certain brain chemicals including acetylcholine and by blocking the movement of certain nerves and muscles. It is not completely known how Botox reduces headache pain and stiffness. A potential reason might be that Botulin Toxin A blocks nerves that send pain messages to the brain and relaxes muscles so they are less sensitive to pain.
Botulin Toxin A’s effectiveness
Patients have differing levels of success with each type of treatment. Some feel reasonably better quickly, while others find their migraine symptoms completely go way for 3-4months.
Who should not take Botulin Toxin A for migraines
People should avoid Botulin Toxin A if they are allergic to any of the ingredients, have had an allergic reaction to other similar products or have a skin infection at the site where Botulin Toxin A will be injected. Caution should be exercised for people who have any disease that affects their muscles or nerves, those with breathing, swallowing or bleeding problems as well as those who have facial weakness such as drooping eyelids or are pregnant/breast feeding women.
Source: www.medcape.com